A Real-World Prospective Study of the Safety and Effectiveness of the Loop Open Source Automated Insulin Delivery System was published on Liebertpub.com, 12 April 2021. This is a very exciting and important study, validating the effectiveness and safety of those of us who have been successfully and happily DIY Looping for many years!
Objective: To evaluate the safety and effectiveness of the Loop Do-It-Yourself automated insulin delivery system.
Conclusions: The Loop open source system can be initiated with community-developed resources and used safely and effectively by adults and children with type 1 diabetes.

Discussion: Open-source AID systems are used by many adults and children to automate insulin delivery for management of T1D, with at least 9000 having used Loop. The study data reflect real-world use of Loop in that there was no guidance provided by the study as to how Loop was to be used and no formal customer support for troubleshooting. The study participants initiated Loop with community-developed resources. TIR, which on average was already at a high level before starting Loop, increased further; and time <54 mg/dL decreased. Improvement in TIR occurred immediately after starting Loop and was sustained on average over 6 months. The benefits of Loop were seen in both adults and children, across the full range of baseline HbA1c, and with both high- and moderate-income levels.
In summary, this real-world study has demonstrated that the Loop open-source hybrid closed-loop system can be safely self-initiated and used by adults and children with T1D and reduced time in range without increasing hypoglycemia. Tidepool is developing a commercial version of Loop (“Tidepool Loop”), which will rely on the data generated in this study to support FDA clearance.
Read the report: A Real-World Prospective Study of the Safety and Effectiveness of the Loop Open Source Automated Insulin Delivery System
Botvinick Wins Grant to Develop Advanced Monitor for Type 1 Diabetes was shared by Anna Lynn Spitzer for UCI.edu/engineering, 6 April 2021.
 UC Irvine’s Elliot Botvinick, professor of biomedical engineering, has been awarded a three-year, $3.5 million grant from The Leona M. and Harry B. Helmsley Charitable Trust to further the development of an innovative continuous-use monitor for those with Type 1 diabetes. The first-of-its-kind device will simultaneously measure insulin, glucose, lactate, oxygen and the ketone body beta-hydroxybutyrate with a single probe inserted just beneath the skin.
UC Irvine’s Elliot Botvinick, professor of biomedical engineering, has been awarded a three-year, $3.5 million grant from The Leona M. and Harry B. Helmsley Charitable Trust to further the development of an innovative continuous-use monitor for those with Type 1 diabetes. The first-of-its-kind device will simultaneously measure insulin, glucose, lactate, oxygen and the ketone body beta-hydroxybutyrate with a single probe inserted just beneath the skin.
Called iGLOBE (Insulin + Glucose + Lactate + Oxygen + Beta-HydroxybutyratE) LifeStrip, the monitor utilizes light and chemistry to provide sensing capabilities for multiple analytes, which can be critical for controlling blood glucose and detecting possible dangerous events in those with the disease. The device will include continuous insulin monitoring and improve dosing efficacy by providing real-time feedback on the dynamics of insulin-pump therapy as well as real-time estimates of a patient’s sensitivity to the insulin.
It is also important to monitor blood glucose in those with Type 1 diabetes, also known as insulin-dependent diabetes. When blood glucose is elevated above normal values, a condition called hyperglycemia, the body produces a chemical called beta-hydroxybutyrate through a metabolic reaction. Elevated beta-hydroxybutyrate is associated with diabetic ketoacidosis, a dangerous condition, which can result in hospitalization or death. iGLOBE monitors this chemical both as a “smoke alarm” to indicate dangerous levels and as an additional test to ensure automated insulin delivery is functioning properly.
Monitoring lactate, produced during exercise, is important as well, as it can indicate changing metabolic states, which can lead to changes in blood glucose in the hours after exercise. Knowledge of such metabolic shifts will improve glucose prediction and improve insulin dosing.
“Clinical evidence suggests that both beta-hydroxybutyrate and insulin sensing would improve outcomes and decrease the rates of hospitalization, severe morbidity and death associated with hypo- and hyperglycemia,” said Botvinick, who is also associate director of UCI’s Edwards Lifesciences Center for Advanced Cardiovascular Technology and a professor of surgery at UCI Beckman Laser Institute (BLI).
Read more: Botvinick Wins Grant to Develop Advanced Monitor for Type 1 Diabetes
Scientists Just Bioprinted a Pancreas That Could End Diabetes was posted by Brad Bergan for InterestingEngineering.com, 20 April 2021. This could also put an end to animal testing.

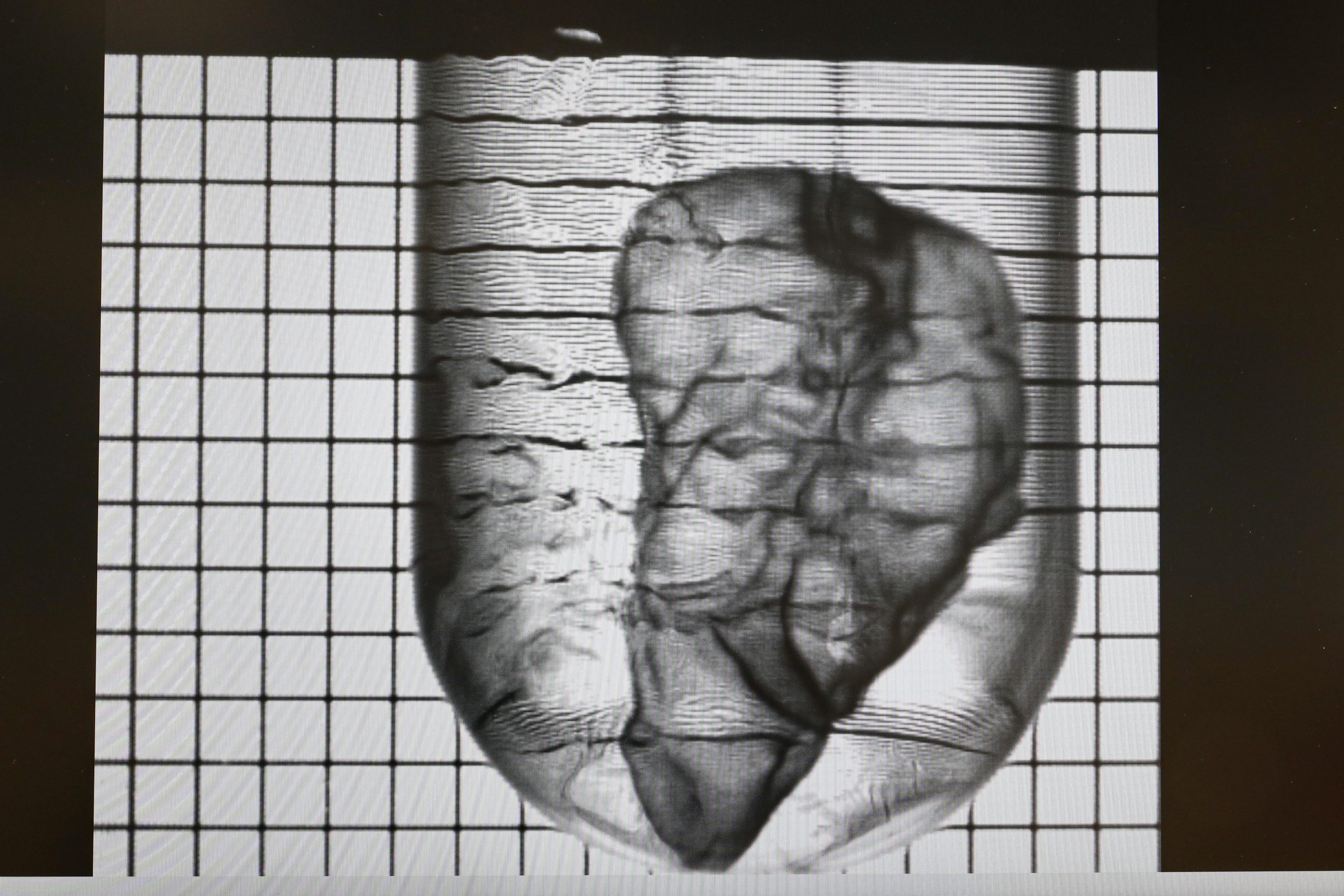 A company recently developed a novel system capable of printing biological tissue in a blindingly fast 30 seconds — creating a possible means of bringing an end to diabetes, according to a blog post shared on the Ecole Polytechnique Federale de Lausanne’s (EPFL’s) official website. Called Readily3D, the company’s technology has seen widespread use in a large-scale European project to build a living model of the human pancreas — which could also provide a safe alternative for testing new drugs. One day, bioprinting technology might even provide viable transplant organs.
A company recently developed a novel system capable of printing biological tissue in a blindingly fast 30 seconds — creating a possible means of bringing an end to diabetes, according to a blog post shared on the Ecole Polytechnique Federale de Lausanne’s (EPFL’s) official website. Called Readily3D, the company’s technology has seen widespread use in a large-scale European project to build a living model of the human pancreas — which could also provide a safe alternative for testing new drugs. One day, bioprinting technology might even provide viable transplant organs.
The benefits are several: a bioprinted pancreas can be totally customized to match a patient’s stem cells — removing the need to conduct animal testing. “What’s more, patients won’t have to try out an array of drugs, some of which may have unpleasant side effects, before finding the right one for them,” said CEO Readily3D Damien Loterie in the report.
Read more: Scientists Just Bioprinted a Pancreas That Could End Diabetes
Medtronic Launches Seven-Day Infusion Set in Europe was reviewed by Albert Cai for diaTribe.org, 19 April 2021. Medtronic’s Extended insulin infusion set doubles the wear-time of a traditional infusion set. The device is launching in Finland and Belgium, with other European countries coming later this year.
 Medtronic recently announced the launch of its new Medtronic Extended infusion set which can be worn for up to seven days. This new infusion set will require fewer site changes and fewer interruptions to insulin delivery, making diabetes management more convenient and less burdensome. Plus, it is designed to maintain insulin flow for longer. The new tubing connector is intended to improve the stability of insulin, the reliability of infusion site performance, and it reduces the risk of infusion set occlusion.
Medtronic recently announced the launch of its new Medtronic Extended infusion set which can be worn for up to seven days. This new infusion set will require fewer site changes and fewer interruptions to insulin delivery, making diabetes management more convenient and less burdensome. Plus, it is designed to maintain insulin flow for longer. The new tubing connector is intended to improve the stability of insulin, the reliability of infusion site performance, and it reduces the risk of infusion set occlusion.
The Medtronic Extended infusion set is compatible with MiniMed 600G and 700G series pumps, including the 670G, 770G, and 780G automated insulin delivery systems. For those in the US, Medtronic expects to submit the Medtronic Extended infusion set to the FDA before the end of this month. A 291-person trial for the device ended in November 2020 and Medtronic plans to share the study results at this year’s ADA 2021 Scientific Sessions.
Read more: Medtronic Launches Seven-Day Infusion Set in Europe
Plastics, sharps diabetes waste require conscious disposal was presented by Michael Monostra for Healio.com/endocrinology, 22 April 2021.
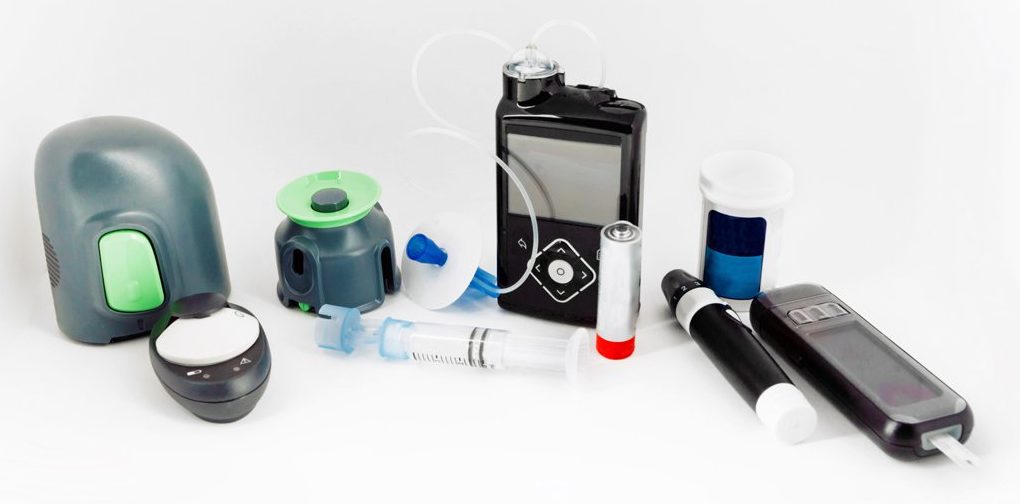 Among the burdens of managing diabetes is responsibly discarding all the packaging, test strips, plastic and sharps. Manufacturers of new technologies are working to reduce waste, but concerns about disposal remain. According to Lucille Hughes, DNP, MSN/ED, CDCES, BC-ADM, FADCES, director of diabetes education at Mount Sinai South Nassau in New York, the advent of continuous glucose monitoring has lessened some of the trash generated by finger sticks, and CGM manufacturers have worked to cut down the amount of plastic used with each new generation to reduce waste further.
Among the burdens of managing diabetes is responsibly discarding all the packaging, test strips, plastic and sharps. Manufacturers of new technologies are working to reduce waste, but concerns about disposal remain. According to Lucille Hughes, DNP, MSN/ED, CDCES, BC-ADM, FADCES, director of diabetes education at Mount Sinai South Nassau in New York, the advent of continuous glucose monitoring has lessened some of the trash generated by finger sticks, and CGM manufacturers have worked to cut down the amount of plastic used with each new generation to reduce waste further.
Bruce Taylor, senior director of government affairs, access and policy for Dexcom, said used sharps disposal has been a challenge government and health officials have tried to tackle for many years. States have regulations for disposing of sharps, but not enough has been done to communicate these rules to patients, Taylor said. “You can pass a law and say you can’t put it in your household trash,” Taylor told Healio. “But unless it is communicated to the patient and the patient understands — how do you safely dispose of sharps, how do you containerize it, how do you safely dispose of it — legislation is going to be completely ineffective.”
This realization led to the founding of SafeNeedleDisposal.org, a website dedicated to helping people safely dispose of used sharps. The website is owned and managed by NeedyMeds, a nonprofit organization serving as an information resource for health care financial assistance programs. Nine health care companies are part of a coalition supporting the website and its mission.
SafeNeedleDisposal.org features a map that users can click to find out the proper way to dispose of sharps in their area. The website advises people to place used sharps in either a sharps disposal bin or a used laundry detergent bottle. Disposal methods are listed for each state, and visitors can put in their ZIP code to find the nearest disposal locations.
Read more: Plastics, sharps diabetes waste require conscious disposal


I am glad our county distributes and collects sharps boxes. I beats toss and hope, my method for 30 years.
By the way why is there so darn much diabetes trash? i hate trash.