Adocia’s Partner Tonghua Dongbao Receives Clearance to Start Ultra-Rapid Insulin BioChaperone Lispro Phase 3 Trial to Treat Type 1 and Type 2 Diabetes in China was published on BioSpace.com, 14 October 2021.
 Adocia, a clinical stage biopharmaceutical company focused on diabetes treatments and other metabolic diseases with innovative formulations of proteins and peptides, announced today that its partner Tonghua Dongbao Pharmaceutical Co. Ltd. received clearance from the Center for Drug Evaluation (CDE) of the China National Medical Products Administration (NMPA) to conduct the Phase 3 clinical trial of BioChaperone Lispro, an Ultra-Rapid Insulin for the treatment of Type 1 and Type 2 diabetes. The first patient enrolled in the program will trigger a milestone payment to Adocia.
Adocia, a clinical stage biopharmaceutical company focused on diabetes treatments and other metabolic diseases with innovative formulations of proteins and peptides, announced today that its partner Tonghua Dongbao Pharmaceutical Co. Ltd. received clearance from the Center for Drug Evaluation (CDE) of the China National Medical Products Administration (NMPA) to conduct the Phase 3 clinical trial of BioChaperone Lispro, an Ultra-Rapid Insulin for the treatment of Type 1 and Type 2 diabetes. The first patient enrolled in the program will trigger a milestone payment to Adocia.
“We are delighted to enter into the Phase 3 program for one of the two products licensed to Tonghua Dongbao, the domestic insulin leader in China,” commented Gerard Soula, President & CEO of Adocia.
 Adocia out-licensed BC Lispro to Tonghua Dongbao who is responsible for clinical development, manufacturing, product registration and future commercial operations in China and other Asian territories. The total amount perceived for this agreement could reach $45m (including $10m upfront payment) and double-digit royalties on sales. In parallel, Adocia is looking for other commercial partners in the US and in Europe for BC Lispro
Adocia out-licensed BC Lispro to Tonghua Dongbao who is responsible for clinical development, manufacturing, product registration and future commercial operations in China and other Asian territories. The total amount perceived for this agreement could reach $45m (including $10m upfront payment) and double-digit royalties on sales. In parallel, Adocia is looking for other commercial partners in the US and in Europe for BC Lispro
Dr. Chunsheng Leng, CEO of Tonghua Dongbao said, “This is an exciting time for us. Based on the strong positive Phase 1 clinical trial results in Europe, delivered by our partner Adocia, we have been able to apply for and now receive CDE clearance to initiate this Phase 3 program. These studies will be run in parallel with the ongoing Phase 1 trial and will greatly shorten our clinical study time. We hope that BC Lispro, this new generation insulin product, will bring a great value for people with diabetes in China, plus the rest of the world.”
In China, the prandial market represents $1.4bn. The fast-acting insulin market segment is the fastest growing with 16% YoY in 2019 (iQVia, May 2021).
Read more: Tonghua Dongbao Receives Clearance to Start Ultra-Rapid Insulin BioChaperone Lispro Phase 3 Trial
Noninvasive Glucose Monitoring: In God We Trust—All Others Bring Data was published by David C. Klonoff, et al in the Journal of Diabetes Science and Technology, 21 October 2021.
 There is great demand for a United States Food and Drug Administration (FDA)-cleared noninvasive glucose monitor. We define this term as a technology where the concentration of glucose is obtained without inserting a device into the body or puncturing the skin. When the measurement involves passing a type of radiation into the body, the technology is referred to as a noninvasive optical glucose monitor (NIO-GM). The amount of interest in seeing the development of an accurate, viable NIO-GM and the amount of hyperbole by companies promising accurate NIO-GMs both far outstrip the amount of publicly available data actually generated by these potential products. This mismatch in the production of large amounts of claims and predictions compared to small amounts of actual data accounts for the title of our editorial.
There is great demand for a United States Food and Drug Administration (FDA)-cleared noninvasive glucose monitor. We define this term as a technology where the concentration of glucose is obtained without inserting a device into the body or puncturing the skin. When the measurement involves passing a type of radiation into the body, the technology is referred to as a noninvasive optical glucose monitor (NIO-GM). The amount of interest in seeing the development of an accurate, viable NIO-GM and the amount of hyperbole by companies promising accurate NIO-GMs both far outstrip the amount of publicly available data actually generated by these potential products. This mismatch in the production of large amounts of claims and predictions compared to small amounts of actual data accounts for the title of our editorial.
Access to an NIO-GM would be very appealing to people with diabetes who want to know their glucose concentrations before and after meals, as well as during other activities throughout the day, so they can titrate their activities and doses of medications without pain, inconvenience, or blood waste. Even people without diabetes are interested in the continuous glucose monitors (CGMs) currently on the market as a way to track dietary intake and improve fitness.
On August 25, 2021, we conducted a Google search for articles published within the previous sixty days using the search phrase “noninvasive glucose.” In the first ten pages (100 articles) we found stories about eleven companies developing NIO-GMs. These companies included Hagar (Tel Aviv, Israel, August 12, 2021), Rockley Photonics (Pasadena, California, July 19, 2021), Apple (Cupertino, California, July 19, 2021), Samsung (Seoul, South Korea, August 15, 2021), Integrity Applications (Ashdod, Israel, August 12, 2021), Meta Materials Inc. (Dartmouth, Nova Scotia, Canada, August 13, 2021), Movano (Pleasanton, California, August 12, 2021), Lucid Diagnostics Inc. (New York, New York, August 10, 2021), Nanoco (Manchester, UK, August 12, 2021), Afon Technology (Caldicot, UK, June 29, 2021), and GBT Technologies Inc. (Santa Monica, California, August 24, 2021). We also found an article about a company with a noninvasive fluid sampling glucose monitor (NIFS-GM) device, which measures glucose concentrations in a fluid collected from the body in a noninvasive manner (i.e., without the need to puncture skin). This company is Nemaura Medical (Loughborough, UK, August 12, 2021). To our knowledge, only one of these twelve companies (Integrity Applications) has published noninvasive glucose data in a peer-reviewed medical journal and the article by Integrity Applications appeared three years ago. In a recently published review article, 28 companies were identified as actively pursuing NIO-GMs. Little clinical data has been reported by any of these companies.
We predict difficulties when these entrepreneurial companies are ready to expand their
-
- test populations to a wide demographic,
- test sites from in-clinic to in-home, and
- range of tested glucose concentrations to extreme levels like those that can occur in the real world.
The data reported by these companies is generally too preliminary to justify the overhyped announcements commonly released from this industry.
 CONCLUSION: The most efficient path for the successful development of clinically viable NIO-GMs is to disseminate hypotheses, protocols, and data within the medical, scientific, engineering, regulatory, and business communities. Products with only secret, unpublished data will ultimately not be embraced by these communities. The publication of results can stimulate new ideas and approaches with the potential to overcome barriers that currently limit technological progress. The publication of data also has the potential to establish benchmarks that can be used to judge progress in the field and to develop established protocols that can be followed by all research teams, thereby permitting meaningful comparisons of findings between different research groups.
CONCLUSION: The most efficient path for the successful development of clinically viable NIO-GMs is to disseminate hypotheses, protocols, and data within the medical, scientific, engineering, regulatory, and business communities. Products with only secret, unpublished data will ultimately not be embraced by these communities. The publication of results can stimulate new ideas and approaches with the potential to overcome barriers that currently limit technological progress. The publication of data also has the potential to establish benchmarks that can be used to judge progress in the field and to develop established protocols that can be followed by all research teams, thereby permitting meaningful comparisons of findings between different research groups.
An iconic line from the movie Jerry Maguire was “Show me the money!” Our approach to noninvasive glucose monitoring, where we read every few weeks about a new breakthrough technology, is “Show me the data!”
Read more: Noninvasive Glucose Monitoring: In God We Trust—All Others Bring Data
Capsinoids, Red Pepper, May Lower Glucose in People Over 40 Years of Age was reported by Jessica Nye PhD for EndocrinologyAdvisor.com, 2 November 2021.
 A systematic review and meta-analysis found inconsistent evidence of the benefits of glycemic control from supplementation with capsinoids. These findings were published in The International Journal of Clinical Practice.
A systematic review and meta-analysis found inconsistent evidence of the benefits of glycemic control from supplementation with capsinoids. These findings were published in The International Journal of Clinical Practice.
Researchers from Tehran University of Medical Sciences in Iran searched publication databases through May of 2020 for studies of capsinoids in the setting of glycemic control.The study authors concluded that there was no clear benefit on glycemic control from supplementation with capsinoids, however, “capsinoids and red pepper significantly reduce glucose in people older than 40 years.”
Very limited and inconclusive … BUT, if you are a T1D over 40, what’s your experience?
Read more: Capsinoids, Red Pepper, May Lower Glucose in People Over 40 Years of Age
Researcher bets the gut hormone GLP-1 can be used to treat far more than diabetes was published by the University of Toronto for MedicalXPress.com, 4 November 2021.
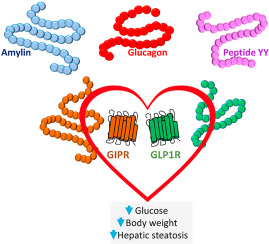 Daniel Drucker, a professor in the department of medicine at the University of Toronto’s Temerty Faculty of Medicine and a senior scientist at the Lunenfeld-Tanenbaum Research Institute at Sinai Health, has pioneered research on gut hormones that has led to life-changing therapies for people with type 2 diabetes, obesity and short bowel syndrome. Now, Drucker’s lab is studying how these same hormones work in the context of other conditions throughout the body, which could result in treatments for an even wider variety of diseases.
Daniel Drucker, a professor in the department of medicine at the University of Toronto’s Temerty Faculty of Medicine and a senior scientist at the Lunenfeld-Tanenbaum Research Institute at Sinai Health, has pioneered research on gut hormones that has led to life-changing therapies for people with type 2 diabetes, obesity and short bowel syndrome. Now, Drucker’s lab is studying how these same hormones work in the context of other conditions throughout the body, which could result in treatments for an even wider variety of diseases.
Drucker, an inductee to the Canadian Medical Hall of Fame and winner of the Canada Gairdner International Award, is most well-known for his contributions to the discovery of glucagon-like peptides (GLP-1 and GLP-2), gut hormones that help control insulin and balance blood sugar levels, and for the development of related therapies for diabetes, obesity and intestinal failure.
Yet, beyond conventional metabolism, drugs based on GLP-1 can also reduce plaque formation in arteries, or atherosclerosis, and control inflammation in several organs. Plaque and inflammation are linked to heart attack, stroke and other cardiovascular diseases—some of the leading causes of death in people with type 2 diabetes and obesity.
The drugs also show promise for treating liver disease and Alzheimer’s disease.
“This paper, recently published in JCI Insight, is the first to show that even though weight loss is the same in both groups of animals that we studied, the animals that were missing the GLP-1 receptor in the immune cells in the liver did not have the same therapeutic benefit,” Drucker says. “It’s really the first paper to show that there’s another element to the story of how GLP-1 works in the liver.”
Read more: Gut hormone GLP-1 can be used to treat far more than diabetes
Hybrid Closed-loop to Manage Gastroparesis in People With Type 1 Diabetes: a Case Series was published in the Journal of Diabetes Science and Technology, 11 August 2021.
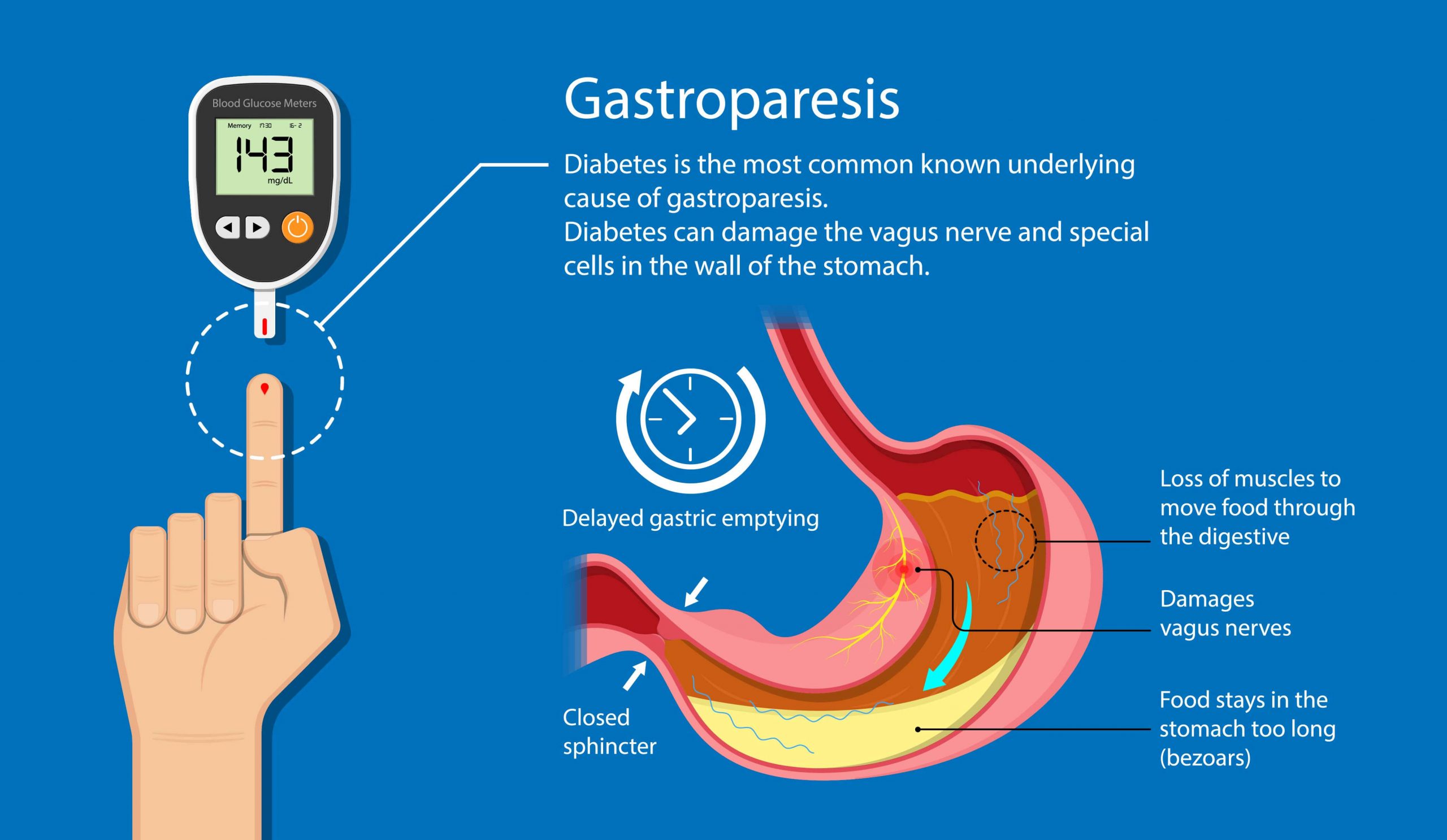 Gastroparesis is associated with unpredictable gastric emptying and can lead to erratic glucose profiles and negative impacts on quality-of-life. Many people with gastroparesis are unable to meet glycemic targets and there is a need for new approaches for this population. Hybrid closed-loop systems improve glucose control and quality-of-life but evidence for their use in people with diabetic gastroparesis is limited.
Gastroparesis is associated with unpredictable gastric emptying and can lead to erratic glucose profiles and negative impacts on quality-of-life. Many people with gastroparesis are unable to meet glycemic targets and there is a need for new approaches for this population. Hybrid closed-loop systems improve glucose control and quality-of-life but evidence for their use in people with diabetic gastroparesis is limited.
The case series of 7 people with type 1 diabetes and gastroparesis. We compare glycemic control before and during the first 12 months of hybrid closed-loop therapy. Five of 7 patients had data available for time in range before and during hybrid closed-loop therapy, and all had an improvement in percentage time in target glucose range
Conclusion: Hybrid closed-loop systems may represent a valuable approach to improve glycemic control for people with type 1 diabetes and gastroparesis.
Read more: Hybrid Closed-loop to Manage Gastroparesis in People With Type 1 Diabetes


Hmm, a pump for Gastroparesis? That sounds a little too far out there. I know that high blood sugar does lead to Gastroparesis. But an insulin pump to treat
Gastroparesis? I think I will stay tuned. But sounds like a great idea.
Your diabetes will vary
rick