
Feb 24, 2025 | Diabetes Business, Featured, Research News, Stories of D Life, Technology Updates
In this week’s issue of The Savvy Diabetic: Aspartame Triggers Insulin Spike Gut bacteria Alters Brain Proteins Rising World Diabetes Prevalence Medtronic Pump Air Travel Warning FTC Case Against PBMs Move Forward Happy Doctors = Practice Autonomy Sage...
Feb 17, 2025 | Diabetes Business, Eating, Featured, Foods, Research News
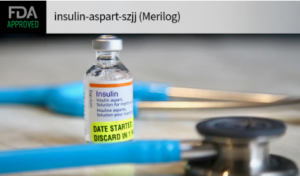
In this week’s issue of The Savvy Diabetic: 1st Rapid-Acting Biosimilar Insulin Approved UVa AI-Powered Insulin Delivery Susvimo for Diabetic Macular Edema Oral Insulin Prevention Trial (PDF) GLP-1 Agonists Increased Risk of Adhesive Capsulitis in Diabetes...

Feb 14, 2025 | Advocacy, Featured
From Breakthrough T1D / JDRF: TAKE ACTION!!! Tell Congress to Protect Critical T1D Research and Cures! Tell Your Members of Congress what we told Secretary Kennedy: there can be no actions that will slow progress toward cures! This is an easy way to make your voice...

Feb 10, 2025 | A Little Humor, Diabetes Business, Featured, Research News, Technology Updates, Videos
In this week’s issue of The Savvy Diabetic: Warning about SmartPhone Alert Settings rBio Addresses Insulin Shortages GlucoTrack Trial Success Yo-Yo Dieting & Kidney Damage Insulin Pump BACKUP Plan (video) Beta Bionics iLet Upgrades (video) Fake...

Feb 3, 2025 | Advocacy, Featured, Foods, Research News
FDA clears use of Ozempic to protect kidney health by Kristin Jensen for BioPharmaDive.com, 29 January 2025. Novo Nordisk won an expanded approval for Ozempic, allowing the company to market its top-selling medicine to reduce the risk of kidney complications in adults...

Jan 27, 2025 | Advocacy, Diabetes Business, Featured, Foods, Research News, Technology Updates, Videos
In this week’s issue of The Savvy Diabetic: Insulin Costs Drop Omnipod Dash Travel Alert FDA At-a-Glance Nutrition Info Modulim & Amputation Research FTC’s Ultimatum to CVS Health ADA & Xeris on Hypoglycemia Education Turmeric & Diabetes...








